| 出品公司: | SBI |
|---|---|
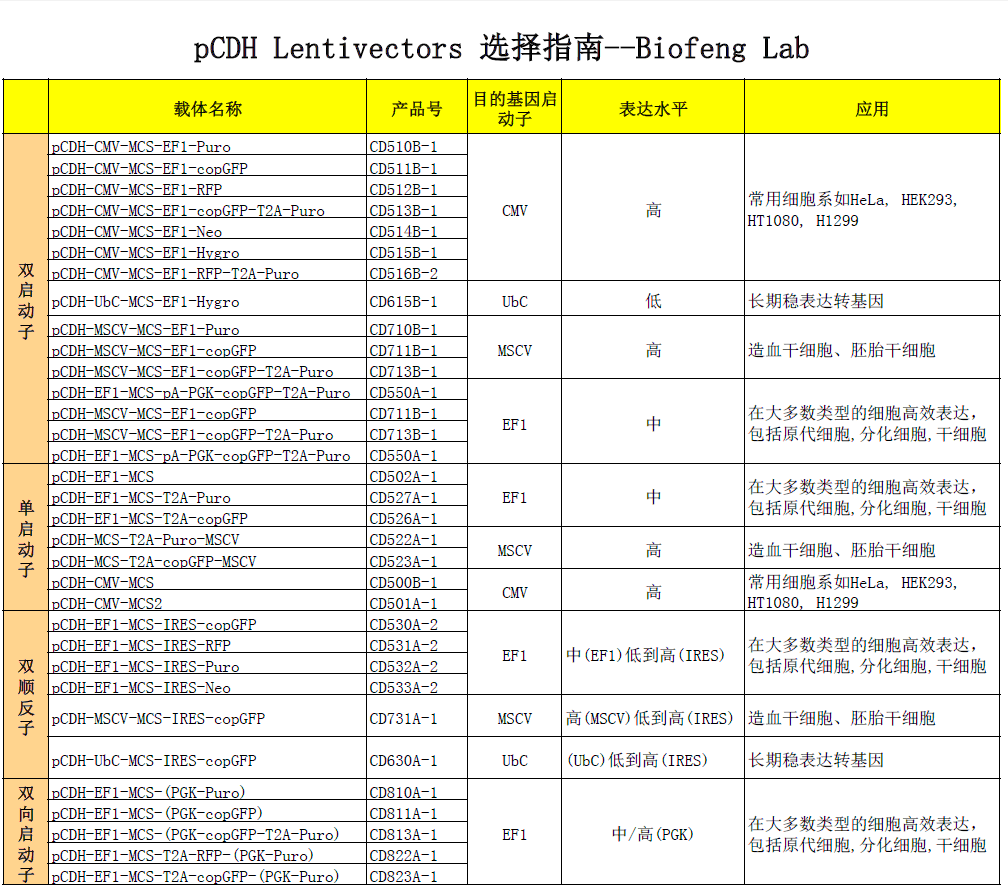
| 载体名称: | pCDH-CMV-MCS-EF1-Hygro, pCDH-CMV-MCS-EF1α-Hygro |
| 质粒类型: | 慢病毒表达载体;cDNA表达载体;双启动子载体 |
| 克隆方法: | 多克隆位点,限制性内切酶 |
| 启动子: | CMV |
| 载体大小: | -- |
| 5' 测序引物及序列: | CMV-F :CGCAAATGGGCGGTAGGCGTG |
| 3' 测序引物及序列: | -- |
| 载体标签: | 无 |
| 载体抗性: | 氨苄青霉素(Ampicillin) |
| 筛选标记: | Hygromycin |
| 克隆菌株: | E.coli cells(RecA-)推荐: Stbl2 ,OmniMAX 2 T1R |
| 宿主细胞(系): | 常用细胞系,如HeLa, HEK293, HT1080, H1299 |
| 备注: |
pCDH-CMV-MCS-EF1-Hygro慢病毒表达载体是基于HIV的慢病毒载体; 用于cDNA表达和克隆;高效转染细胞,建立稳定细胞系; CMV启动子驱动目的基因的高水平表达,EF1a启动子驱动报告基因的中等水平 的表达。 |
| 产品目录号: | CD515B-1 |
| 稳定性: | 稳表达 |
| 组成型/诱导型: | 组成型 |
| 病毒/非病毒: | 慢病毒(HIV) |
买家导航

pCDH-CMV-MCS-EF1-Hygro载体 pCDH-CMV-MCS-EF1α-Hygro质粒基本信息
pCDH-CMV-MCS-EF1-Hygro质粒图谱 pCDH-CMV-MCS-EF1α-Hygro载体图谱和pCDH-CMV-MCS-EF1-Hygro载体序列 pCDH-CMV-MCS-EF1α-Hygro质粒序列多克隆位点信息
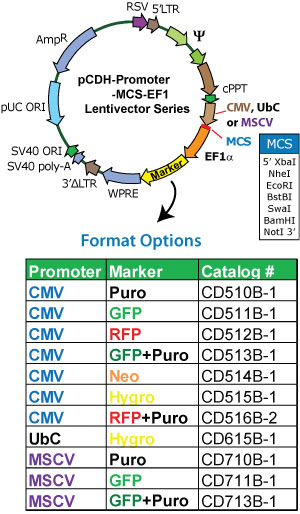
pCDH-CMV-MCS-EF1-Hygro质粒 pCDH-CMV-MCS-EF1α-Hygro载体简介
背景简介:
This manual provides details and information necessary to generate expression constructs of your gene of interest in the pCDH cDNA Cloning and Expression Lentivectors. Specifically, it provides critical instructions on amplification and cloning cDNA into the pCDH vectors, and verification of the final expression constructs. This manual does not include information on packaging the pCDH expression constructs into pseudotyped viral particles or transducing your target cells of choice with these particles. This information is available in the user manual Lentivector Expression Systems: Guide to Packaging and Transduction of Target Cells which is available on the SBI website. Before using the reagents and material supplied with this system, please read the entire manual.
基于HIV-1的pCDH 慢病毒载体特征:
Multiple Cloning Site (MCS)—for cloning the gene of interest in the MCS located downstream of the CMV promoter.
WPRE element—enhances stability and translation of the CMV-driven transcripts.
SV40 polyadenylation signal—enables efficient termination of transcription and processing of recombinant transcripts.
Hybrid RSV/5LTR promoter—provides a high level of expression of the full-length viral transcript in producer 293 cells.
Genetic elements (cPPT, gag, env, LTRs)—necessary for packaging, transducing, and stably integrating the vira expression construct into genomic DNA.
SV40 origin—for stable propagation of the pCDH plasmid in mammalian cells.
pUC origin—for high copy replication and maintenance of the plasmid in E.coli cells.
Ampicillin resistance gene—for selection in E.coli cells.
pCDH 慢病毒表达载体的优势:
Lentiviral expression vectors are the most effective vehicles for the delivery and expression of a gene of interest to almost any mammalian cell—including non-dividing cells and model organisms (C.A. Machida, 2003; M. Federico, 2003; W. C. Heiser, 2004). As with standard plasmid vectors, it is possible to introduce lentivector expression constructs in plasmid form into the cells with low-to-medium efficiency using conventional transfection protocols. However, by packaging the lentivector construct into viral particles, you can obtain highly efficient transduction of expression constructs—even with the most difficult to transfect cells, such as primary, stem, and differentiated cells. The expression construct transduced in target cells is integrated into genomic DNA and provides stable, long-term expression of the target gene.
pCDH 慢病毒载体的包装载体及细胞系
The expression lentivector contains the genetic elements responsible for packaging, transduction, stable integration of the viral expression construct into genomic DNA, and expression of the target gene sequence. The packaging vector provides all the proteins essential for transcription and packaging of an RNA copy of the expression construct into recombinant viral particles. To produce a high titer of viral particles, expression and packaging vectors are transiently co-transfected into producer mammalian cells (e.g., HEK 293 cells). For a detailed description of SBI’s Lentivector expression system,please refer to the Lentivector Expression System user manual.
启动子的选择:
SBI provides a collection of cDNA cloning and expression vectors for various applications. A gene of interest can be cloned under a CMV or EF1 promoter with or without another expression cassette for a reporter gene (copGFP or PuroR). Genes can be either expressed transiently through transfection or stably expressed in a target cell line through transduction with packaged viral particles.
The major concern of cDNA expression in lentivectors is the efficiency level and stability of expression in target cell lines.
The Cytomegalovirus (CMV) promoter is a strong and most commonly used viral promoter that constitutively expresses downstream genes. While the CMV promoter works perfectly in the most common cell lines, it shows poor expression in some stem cell lines and hematopoietic cell lines (R.F. Doll, 1996; E.D. Papadakis, 2004).
The housekeeping elongation factor 1α (EF1) promoter has been shown to exceed and outlast CMV-mediated expression in retroviral, lentiviral, and adenoviral vectors, in hematopoietic cell lines (K. Tokushige 1997; H. Nakai, 1998; C. Teschendorf, 2002). EF1 also performs well in most common cell lines.
MSCV promoter is the 5’-LTR promoter of murine stem cell virus. When a portion of the U3 region of the 3’ HIV LTR was replaced with the U3 region of MSCV LTR, the resulted hybrid HIV/MSCV LTR has dramatically increased the transgene expression level in human CD34+ hematopoietic cells (J.K. Choi, 2001). After integration into genomic DNA, this promoter transcribes a long transcript with an intron in the 5’UTR flanked with splice donor and acceptor sites derived from the lentiviral vector. Further studies found that additional CpG mutations in the MSCV LTR reduced transcriptional silencing in embryonic stem cells (C.S. Swindle, 2004). We constructed cDNA expression vectors with the CpG-deficient MSCV incorporated into the 3’ HIV LTR. After integration into genomic DNA, 3’MSCV/LTR will replace the 5’LTR and provide a high level of expression of the target gene and reporter gene downstream.
SBI第三代慢病毒载体
SBI offers a third generation of the most popular HIV-1 based lentivector expression system which consists of three maincomponents:
(1) The lentiviral expression vector (e.g., pCDH-EF1-MCS-T2A-Puro)
(2) The lentiviral packaging plasmids (e.g., pPACKH1 Packaging Plasmid mix)
(3) A pseudoviral particle producer cell line (e.g., 293TN cells)
2A Peptide-enabled dual expression system
Coexpression of a reporter gene together with a gene of interest is a useful approach for selecting transfected or transduced cells. This is commonly achieved by using two independent internal promoters, such as CMV and EF1 in pCDH-CMV-MCSEF1- copGFP, or by linking two transgenes with an internal ribosomal entry site (IRES) element in a single bicistronic transcript. Many dual promoter pairs have shown a high level of expression of both transgenes in standard cell lines— however, promoter interference often occurs in some cell lines. There are also two main problems that limit the use of IRES: the large size and the imbalanced expression between the first and second cistrons (H. Mizuguchi, 2000; X.Yu, 2003).
The “self-cleaving” 2A peptides have been used successfully to generate multiple proteins from a single promoter in many applications (P. de Felipe, 2004; M.J. Osborn, 2005; P. de Felipe, 2006). The 2A-like sequences exist in several viruses and are used to mediate protein cleavage from a single open reading frame. Through a ribosomal skip mechanism, the 2A peptide prevents normal peptide bond formation between the 2A glycine and the 2B proline without affecting the translation of 2B (M.L. Donnelly, 2001):SBI’s cDNA expression vectors incorporate the 2A-like sequence (T2A) from the insect virus Thosea asigna to mediate the coexpression of a reporter gene with the target cDNA. Reporter genes have been cloned at either the first or second positions, and we achieved high expression levels at both locations.