| 出品公司: | Imgenex |
|---|---|
| 载体名称: | pCL-Ampho, pCL Ampho |
| 质粒类型: | 哺乳动物载体;逆病毒包装;单质粒包装系统 |
| 高拷贝/低拷贝: | 高拷贝 |
| 克隆方法: | 限制性内切酶,多克隆位点 |
| 启动子: | CMV |
| 载体大小: | 约11kb |
| 5' 测序引物及序列: | CMV fwd 5’CGCAAATGGGCGGTAGGCGTG 3’ |
| 3' 测序引物及序列: | -- |
| 载体标签: | 无 |
| 载体抗性: | Ampicillin |
| 筛选标记: | 无 |
| 克隆菌株: | DH5α或 HB101 |
| 宿主细胞(系): | 包装细胞系如293T |
| 备注: |
逆病毒包装载pCL-Ampho是单质粒包装系统,快速产生Helper-free、毒力高的逆病毒颗粒。 可以用于大部分哺乳动物细胞,但是不可以用于仓鼠细胞。 同类质粒有 pCL-Eco 、 pCL-10A 。 |
| 产品目录号: | 10046p/ 10040K |
| 稳定性: | 瞬表达 |
| 组成型: | 组成型 |
| 病毒/非病毒: | 非病毒 |
买家导航

pCL-Ampho载体质粒基本信息
pCL-Ampho质粒图谱载体图谱和pCL-Ampho载体序列质粒序列多克隆位点信息
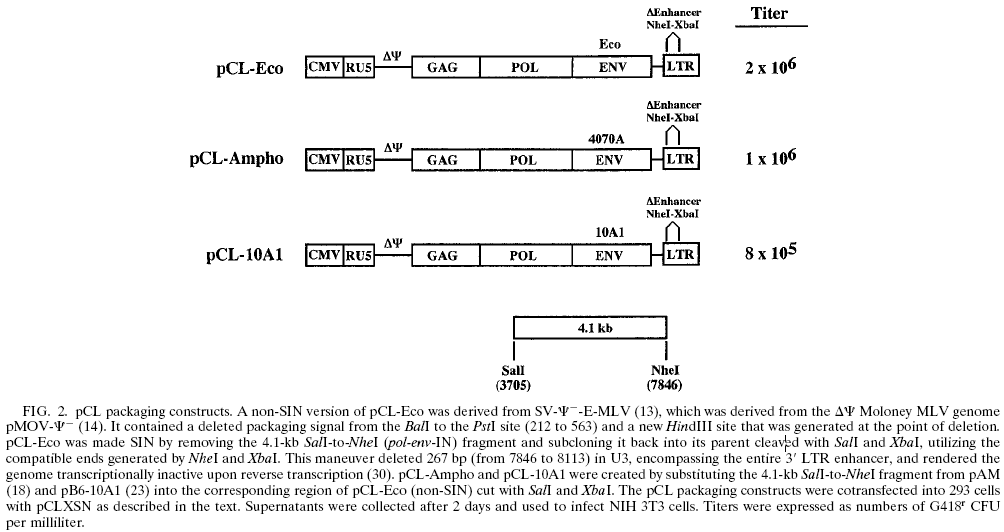
pCL-Ampho质粒载体简介
逆病毒包装载体pCL-Ampho的使用方法——逆病毒包装与转染方法
293T Growth Medium:
450 ml DMEM (JRH Biosciences)
50 ml FBS (JRH Sciences)
5 ml Glutamine (200 mM in saline, JRH Sciences)
2.5 ml Gentamycin (10 mg/ml in water, Sigma)
HC11 Growth Medium:
5 ml L-Glutamine (200 mM in saline, JRH Sciences)
2.5 ml Bovine Insulin (1 mg/ml in 0.01 N HCL, Sigma)
2.5 ml Gentamycin (10 mg/ml in water, Sigma)
0.5 ml EGF (10 μg/ml in water, Gibco)
50 ml FBS (JRH Sciences)
440 ml RPMI (JRH Sciences)
Day 0: Split 293T cells for transfection:
Wash cells off plate with 10 ml Hanks and put into 15 ml Falcon tube
Add 1 ml 10X trypsin and mix by inversion for about 1-2 minutes
Spin at 1000 rpm in clinical centrifuge for 1-2 minutes
Resuspend cells in 10 ml growth medium
Count cells
Plate cells at 1X106 cells per 100 mm dish
Day 2: Transfect 293T cells:
Plan transfection using 2 μg total DNA and 12 μl FuGene in 200 μl total
volume per 100 mm dish
Dilute Fugene: 12 μl per reaction in serum free DMEM for total volume of
200 μl
Mix DNA plasmids in polypropelene tubes
(Falcon #2063):
1 μg DNA of interest
1 μg pCL-Ampho
Add 200 μl diluted FuGene dropwise to each DNA tube
Incubate RT 15 min
Feed cells with 5 ml fresh growth media and add FuGene/DNA mixture to media
Day 3: Change medium:
• Remove viral medium and CAREFULLY feed with 6 ml growth medium
This step may reduce a cytostatic factor (produced after transfections) that
can inhibit growth and infection of your target cells.
Split HC11 cells for infection
Seed HC11 cells at 300,000 cells per 100 mm dish
If using coverslips, incubate with FBS for at least 1 hour prior to plating
Day 4: Infect target (HC11) cells:
• Collect virus-containing media off 293T cells
• Syringe filter media through .45 μm filter onto target cells
(Evaporation O/N and loss in filter leaves about 5 ml per plate)
• Add 10 μl 5mg/ml polybrene (final conc = 10 μg polybrene/ml media)
(Store polybrene in aliquots to decrease freeze/thaw cycles)
• Wrap plates in parafilm
• Spin cells in clinical centrifuge 10 min at 1800 rpm
• Rotate plate 1/3. Spin 10 min at 1800 rpm
• Rotate plate 1/3. Spin 10 min at 1800 rpm
• Remove viral media and replace with fresh growth media
Day 6+: Harvest HC11 cells:
• Wash cells with HBSS
• Scrape remaining cells, pellet and flash freeze for protein/RNA analysis
Stain cells for beta-gal expression:
Fixing Solution :
1.35 ml 37% Formaldehyde
0.2 ml Gluteraldehyde
1X PBS to 25 mls
Staining Solution:
625μl 40 mg/ml X-gal in DMF
50 μl 1M MgCL2
750 μl 100 mM Potassium Ferricyanide
750 μl 100 mM Potassium Ferrocyanide
1X PBS to 25 mls
[100 mM Potassium Ferrocyanide = 2.11 g/50ml PBS]
[100 mM Potassium Ferricyanide = 1.64 g/50 ml PBS]
Store at room temperature, protect from light.
1. Make -gal solution and warm in 37°C water bath to prevent crystallization
2. Aspirate off media
3. Wash cells 1X PBS
4. Fix cells 5 min at RT in fixing solution
5. Wash 3X PBS
6 Optional: Add PBS to cells and warm plate at 37°C 5 min (reduces
crystallization)
7. Stain cells with staining solution at 37°C for 1-24 hrs
8. Store stained cells in 70% EtOH at 4°C
(EMBO J. 5: pg 3133, 1986)
Note:
HC11 and MEC primary culture cells are very adherent and can withstand
the force of spinning the plates at 1800 rpm. We found that HC11 cells died at
2200 rpm. However, this is cell type-dependent, and spinning speed should be
optimized accordingly.
Expect to see -gal staining at the periphery of the plate. During the spin
infection the media is pushed to the outside. The cells in the center of the plate
will not be exposed to virus and will not be efficiently infected with retrovirus.
pCL-Ampho质粒序列载体序列
pCL-Ampho载体图谱质粒图谱pdf版和pCL-Ampho载体序列质粒序列等相关资料下载
pCL-Ampho质粒载体应用举例
- 上一篇:pBABE-Puro
- 下一篇:pCMV-VSVG

