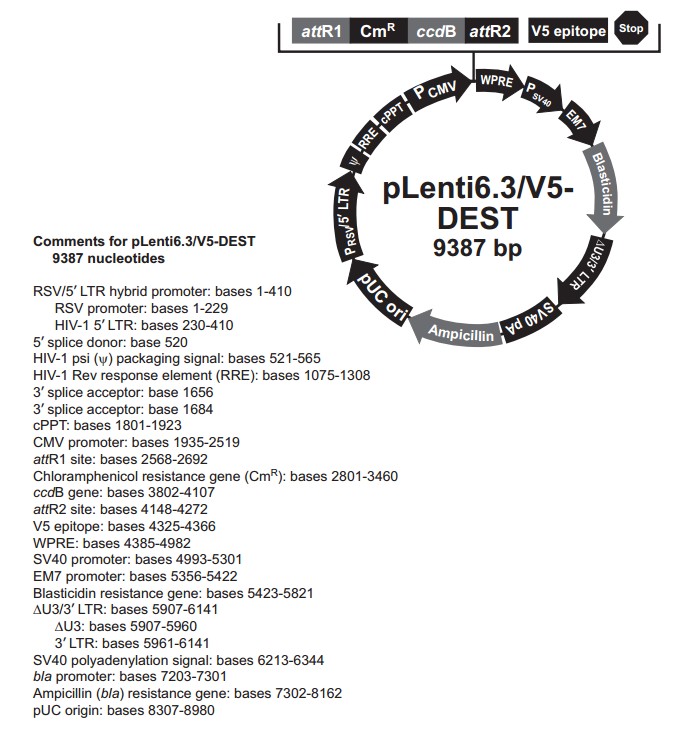
pLenti6.3/V5-DEST载体描述
Invitrogen's pLenti6.3⁄V5-DEST Gateway Vector Kit is part of our ViraPower HiPerform Lentiviral Gateway Expression Kit .
The pLenti6.3⁄V5-DEST Gateway Vector Kit contains the Gateway-adapted ViraPower HiPerform lentiviral expression vector, pLenti6.3⁄V5-DEST for easy recombination-based cloning and high-level expression of a target gene in dividing and non-dividing mammalian cells. The pLenti6.3⁄V5-DEST vector is equipped with two key genetic elements, making it a HiPerform vector: the Woodchuck Posttranscriptional Regulatory Element (WPRE) and the central Polypurine Tract (cPPT) sequence from the HIV-1 integrase gene to produce at least 4-fold increase in protein expression compared to vectors lacking these elements.
pLenti6.3/V5-DEST载体优点
• Stable expression
• Long-term experiments
• Accurate titer of functional virus
• Flexible and versatile Gateway® recombination cloning technology
pLenti6.3/V5-DEST载体特征
• HiPerform™ WPRE and cPPT elements
• CMV promoter
• V5 epitope tag at C terminus
• Blasticidin selection
Gateway技术
The Gateway Technology is a universal cloning method that takes advantage of the site-specific recombination properties of bacteriophage lambda (Landy, 1989) to provide a rapid and highly efficient way to move your gene of interest into multiple vector systems. To express your gene of interest using
Gateway Technology, simply:
1. Generate entry clones containing your promoter and gene(s) of interest.
2. Generate an expression clone by performing an LR recombination reaction between the entry clone(s) and pLenti6.3/V5-DEST).
3. Transfect your expression clone into cells of your choice to transiently or stably express your gene of interest.
To fit all of your expression needs, Invitrogen offers state-of-the-art Gateway destination vectors for expression in E. coli, insect, yeast, or mammalian cells, as well as for production of native protein or N- or C-terminal fusion proteins. All Gateway destination vectors have attR sites for recombination with any attL-flanked fragment, regardless of whether it is an entry clone or an Ultimate ORF Clone. The following table lists a variety of available destination vectors.
Additional materials required, available separately: Gateway entry clone, appropriate Gateway LR Clonase enzyme mix, and reaction buffer.
所需材料
在着手开始实验前你需要准备一下材料:
Gateway entry clone, appropriate Gateway LR Clonase enzyme mix, and reaction buffer.
• Purified plasmid DNA of your entry clone(s) (10 fmoles each)
• pLenti6.3/V5-DEST (20 fmoles)
• LR Clonase II Plus enzyme mix (keep at –20°C until immediately before use)
• 1X TE Buffer, pH 8.0 (10 mM Tris-HCl, pH 8.0, 1 mM EDTA)
• 2 μg/μL Proteinase K solution (supplied with the enzyme mix; thaw and keep on ice until use)
• Appropriate competent E. coli host and growth media for expression
• S.O.C. Medium
• LB agar plates containing 100 μg/mL ampicillin
进行LR重组反应
值得注意的事项:
If you use E. coli cells with a transformation efficiency of ≥1 × 108 cfu/μg, a typical LR reaction should give >5,000 colonies if the entire reaction is transformed and plated.
For multiple fragment reactions, typical numbers of colonies (per 10 μL LR reaction) are:
• 2-fragment recombination reaction: 2,000–15,000
• 3-fragment recombination reaction: 1,000–5,000
• 4-fragment recombination reaction: 50–500
Confirming the Expression Clone
The ccdB gene mutates at a very low frequency, resulting in a very low number of false positives. True expression clones will be ampicillin-resistant and chloramphenicol-sensitive. Transformants containing a plasmid with a mutated ccdB gene will be both ampicillin- and chloramphenicol-resistant.
To check your putative expression clone, test for growth on LB plates containing 30 μg/mL chloramphenicol. A true expression clone will not grow in the presence of chloramphenicol.
Sequencing To confirm that your gene of interest is in frame with the C-terminal V5 epitope, you may sequence your expression construct, if desired. We suggest using the following primer sequences.
LOCUS pLenti6.3/V5-DEST 9387 bp DNA SYN
DEFINITION pLenti6.3/V5-DEST
ACCESSION
KEYWORDS
SOURCE
ORGANISM other sequences; artificial sequences; vectors.
FEATURES Location/Qualifiers
source 1..9387
/organism="pLenti6.3/V5-DEST"
/mol_type="other DNA"
misc_feature 230..410
/label="truncHIV-1_3_LTR"
misc_feature 230..410
/label="HIV-1_5_LTR"
misc_feature 521..565
/label="HIV-1_psi_pack"
CDS 953..1963
/label="ORF frame 2"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 1075..1308
/label="RRE"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 1808..1823
/label="cPPT"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
promoter 1941..2445
/label="CMV_immearly_promoter"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 1948..2235
/label="CAG_enhancer"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 2402..2422
/label="CMV_fwd_primer"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
promoter 2403..2472
/label="CMV_promoter"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 2446..2465
/label="pCEP_fwd_primer"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 2448..2472
/label="LNCX_primer"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature 2568..2692
/label="attR1"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
misc_feature complement(2592..2692)
/label="attR2"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
promoter 2717..2740
/label="lac_promoter"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
gene 2801..3460
/label="CAT/CamR"
/gene="CAT/CamR"
/translation="MRDNWRSELYKYKVVKIEPLGVAPTKAKRRVVQREKRAVGIGAL
FLGFLGAAGSTMGAASMTLTVQARQLLSGIVQQQNNLLRAIEAQQHLLQLTVWGIKQL
QARILAVERYLKDQQLLGIWGCSGKLICTTAVPWNASWSNKSLEQIWNHTTWMEWDRE
INNYTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWFNITNWLWYIKLFIMIVGGL
VGLRIVFAVLSIVNRVRQGYSPLSFQTHLPTPRGPDRPEGIEEEGGERDRDRSIRLVN
GSRRYRLTFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFIDKLGSSALHN
LR*"
CDS 2801..3460
/label="ORF frame 2"
/translation="MEKKITGYTTVDISQWHRKEHFEAFQSVAQCTYNQTVQLDITAF
LKTVKKNKHKFYPAFIHILARLMNAHPEFRMAMKDGELVIWDSVHPCYTVFHEQTETF
SSLWSEYHDDFRQFLHIYSQDVACYGENLAYFPKGFIENMFFVSANPWVSFTSFDLNV
ANMDNFFAPVFTMGKYYTQGDKVLMPLAIQVHHAVCDGFHVGRMLNELQQYCDEWQGG
A*"
misc_feature 3802..4107
/label="ccdB"
/translation="MEKKITGYTTVDISQWHRKEHFEAFQSVAQCTYNQTVQLDITAF
LKTVKKNKHKFYPAFIHILARLMNAHPEFRMAMKDGELVIWDSVHPCYTVFHEQTETF
SSLWSEYHDDFRQFLHIYSQDVACYGENLAYFPKGFIENMFFVSANPWVSFTSFDLNV
ANMDNFFAPVFTMGKYYTQGDKVLMPLAIQVHHAVCDGFHVGRMLNELQQYCDEWQGG
A*"
misc_feature complement(4148..4255)
/label="attR1"
/translation="MEKKITGYTTVDISQWHRKEHFEAFQSVAQCTYNQTVQLDITAF
LKTVKKNKHKFYPAFIHILARLMNAHPEFRMAMKDGELVIWDSVHPCYTVFHEQTETF
SSLWSEYHDDFRQFLHIYSQDVACYGENLAYFPKGFIENMFFVSANPWVSFTSFDLNV
ANMDNFFAPVFTMGKYYTQGDKVLMPLAIQVHHAVCDGFHVGRMLNELQQYCDEWQGG
A*"
misc_feature 4148..4248
/label="attR2"
/translation="MEKKITGYTTVDISQWHRKEHFEAFQSVAQCTYNQTVQLDITAF
LKTVKKNKHKFYPAFIHILARLMNAHPEFRMAMKDGELVIWDSVHPCYTVFHEQTETF
SSLWSEYHDDFRQFLHIYSQDVACYGENLAYFPKGFIENMFFVSANPWVSFTSFDLNV
ANMDNFFAPVFTMGKYYTQGDKVLMPLAIQVHHAVCDGFHVGRMLNELQQYCDEWQGG
A*"
CDS 4381..5139
/label="ORF frame 1"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 4392..4967
/label="WPRE"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
promoter 4993..5261
/label="SV40_promoter"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature complement(4993..5196)
/label="SV40_enhancer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
rep_origin 5160..5237
/label="SV40_origin"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5222..5241
/label="SV40pro_F_primer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
promoter 5355..5422
/label="EM7_promoter"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
gene 5423..5821
/label="blast"
/gene="blast"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5891..5912
/label="U3PPT"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5891..5906
/label="cPPT"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5908..5960
/label="delta_U3"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5961..6141
/label="HIV-1_5_LTR"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 5961..6141
/label="truncHIV-1_3_LTR"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
terminator 6216..6335
/label="SV40_PA_terminator"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 6304..6323
/label="EBV_rev_primer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
rep_origin 6353..6451
/label="SV40_origin"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 6436..6455
/label="SV40pro_F_primer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
promoter complement(6530..6548)
/label="T7_promoter"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
promoter complement(6558..6574)
/label="M13_forward20_primer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature complement(6567..6589)
/label="M13_pUC_fwd_primer"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
misc_feature 6567..6715
/label="lacZ_a"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
rep_origin 6733..7039
/label="f1_origin"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
promoter 7232..7260
/label="AmpR_promoter"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
gene 7302..8162
/label="Ampicillin"
/gene="Ampicillin"
/translation="MIDNQPLDYKICERLTGILNYVAPFTLCGYAALMPLYHAIASRM
AFIFSSLYKSWLLSLYEELWPVVRQRGVVCTVFADATPTGWGIATTCQLLSGTFAFPL
PIATAELIAACLARCWTGARLLGTDNSVVLSGKLTSFPWLLACVATWILRGTSFCYVP
SALNPADLPSRGLLPALRPLPRLRLRPQTSRISLWAASPPGDGTGVESPQAPQQAEVC
KACISISQQPGVESPQAPQQAEVCKACISISQQP*"
CDS 7302..8162
/label="ORF frame 3"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
rep_origin 8317..8936
/label="pBR322_origin"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
promoter 9245..9274
/label="lac_promoter"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
misc_feature 9288..9310
/label="M13_pUC_rev_primer"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
promoter 9309..9327
/label="M13_reverse_primer"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
promoter 9344..9363
/label="T3_promoter"
/translation="MSIQHFRVALIPFFAAFCLPVFAHPETLVKVKDAEDQLGARVGY
IELDLNSGKILESFRPEERFPMMSTFKVLLCGAVLSRIDAGQEQLGRRIHYSQNDLVE
YSPVTEKHLTDGMTVRELCSAAITMSDNTAANLLLTTIGGPKELTAFLHNMGDHVTRL
DRWEPELNEAIPNDERDTTMPVAMATTLRKLLTGELLTLASRQQLIDWMEADKVAGPL
LRSALPAGWFIADKSGAGERGSRGIIAALGPDGKPSRIVVIYTTGSQATMDERNRQIA
EIGASLIKHW*"
ORIGIN
1 AATGTAGTCT TATGCAATAC TCTTGTAGTC TTGCAACATG GTAACGATGA GTTAGCAACA
61 TGCCTTACAA GGAGAGAAAA AGCACCGTGC ATGCCGATTG GTGGAAGTAA GGTGGTACGA
121 TCGTGCCTTA TTAGGAAGGC AACAGACGGG TCTGACATGG ATTGGACGAA CCACTGAATT
181 GCCGCATTGC AGAGATATTG TATTTAAGTG CCTAGCTCGA TACATAAACG GGTCTCTCTG
241 GTTAGACCAG ATCTGAGCCT GGGAGCTCTC TGGCTAACTA GGGAACCCAC TGCTTAAGCC
301 TCAATAAAGC TTGCCTTGAG TGCTTCAAGT AGTGTGTGCC CGTCTGTTGT GTGACTCTGG
361 TAACTAGAGA TCCCTCAGAC CCTTTTAGTC AGTGTGGAAA ATCTCTAGCA GTGGCGCCCG
421 AACAGGGACT TGAAAGCGAA AGGGAAACCA GAGGAGCTCT CTCGACGCAG GACTCGGCTT
481 GCTGAAGCGC GCACGGCAAG AGGCGAGGGG CGGCGACTGG TGAGTACGCC AAAAATTTTG
541 ACTAGCGGAG GCTAGAAGGA GAGAGATGGG TGCGAGAGCG TCAGTATTAA GCGGGGGAGA
601 ATTAGATCGC GATGGGAAAA AATTCGGTTA AGGCCAGGGG GAAAGAAAAA ATATAAATTA
661 AAACATATAG TATGGGCAAG CAGGGAGCTA GAACGATTCG CAGTTAATCC TGGCCTGTTA
721 GAAACATCAG AAGGCTGTAG ACAAATACTG GGACAGCTAC AACCATCCCT TCAGACAGGA
781 TCAGAAGAAC TTAGATCATT ATATAATACA GTAGCAACCC TCTATTGTGT GCATCAAAGG
841 ATAGAGATAA AAGACACCAA GGAAGCTTTA GACAAGATAG AGGAAGAGCA AAACAAAAGT
901 AAGACCACCG CACAGCAAGC GGCCGCTGAT CTTCAGACCT GGAGGAGGAG ATATGAGGGA
961 CAATTGGAGA AGTGAATTAT ATAAATATAA AGTAGTAAAA ATTGAACCAT TAGGAGTAGC
1021 ACCCACCAAG GCAAAGAGAA GAGTGGTGCA GAGAGAAAAA AGAGCAGTGG GAATAGGAGC
1081 TTTGTTCCTT GGGTTCTTGG GAGCAGCAGG AAGCACTATG GGCGCAGCGT CAATGACGCT
1141 GACGGTACAG GCCAGACAAT TATTGTCTGG TATAGTGCAG CAGCAGAACA ATTTGCTGAG
1201 GGCTATTGAG GCGCAACAGC ATCTGTTGCA ACTCACAGTC TGGGGCATCA AGCAGCTCCA
1261 GGCAAGAATC CTGGCTGTGG AAAGATACCT AAAGGATCAA CAGCTCCTGG GGATTTGGGG
1321 TTGCTCTGGA AAACTCATTT GCACCACTGC TGTGCCTTGG AATGCTAGTT GGAGTAATAA
1381 ATCTCTGGAA CAGATTTGGA ATCACACGAC CTGGATGGAG TGGGACAGAG AAATTAACAA
1441 TTACACAAGC TTAATACACT CCTTAATTGA AGAATCGCAA AACCAGCAAG AAAAGAATGA
1501 ACAAGAATTA TTGGAATTAG ATAAATGGGC AAGTTTGTGG AATTGGTTTA ACATAACAAA
1561 TTGGCTGTGG TATATAAAAT TATTCATAAT GATAGTAGGA GGCTTGGTAG GTTTAAGAAT
1621 AGTTTTTGCT GTACTTTCTA TAGTGAATAG AGTTAGGCAG GGATATTCAC CATTATCGTT
1681 TCAGACCCAC CTCCCAACCC CGAGGGGACC CGACAGGCCC GAAGGAATAG AAGAAGAAGG
1741 TGGAGAGAGA GACAGAGACA GATCCATTCG ATTAGTGAAC GGATCTCGAC GGTATCGGTT
1801 AACTTTTAAA AGAAAAGGGG GGATTGGGGG GTACAGTGCA GGGGAAAGAA TAGTAGACAT
1861 AATAGCAACA GACATACAAA CTAAAGAATT ACAAAAACAA ATTACAAAAA TTCAAAATTT
1921 TATCGATAAG CTTGGGAGTT CCGCGTTACA TAACTTACGG TAAATGGCCC GCCTGGCTGA
1981 CCGCCCAACG ACCCCCGCCC ATTGACGTCA ATAATGACGT ATGTTCCCAT AGTAACGCCA
2041 ATAGGGACTT TCCATTGACG TCAATGGGTG GAGTATTTAC GGTAAACTGC CCACTTGGCA
2101 GTACATCAAG TGTATCATAT GCCAAGTACG CCCCCTATTG ACGTCAATGA CGGTAAATGG
2161 CCCGCCTGGC ATTATGCCCA GTACATGACC TTATGGGACT TTCCTACTTG GCAGTACATC
2221 TACGTATTAG TCATCGCTAT TACCATGGTG ATGCGGTTTT GGCAGTACAT CAATGGGCGT
2281 GGATAGCGGT TTGACTCACG GGGATTTCCA AGTCTCCACC CCATTGACGT CAATGGGAGT
2341 TTGTTTTGGC ACCAAAATCA ACGGGACTTT CCAAAATGTC GTAACAACTC CGCCCCATTG
2401 ACGCAAATGG GCGGTAGGCG TGTACGGTGG GAGGTCTATA TAAGCAGAGC TCGTTTAGTG
2461 AACCGTCAGA TCGCCTGGAG ACGCCATCCA CGCTGTTTTG ACCTCCATAG AAGACACCGA
2521 CTCTAGAGGA TCCACTAGTC CAGTGTGGTG GAATTCTGCA GATATCAACA AGTTTGTACA
2581 AAAAAGCTGA ACGAGAAACG TAAAATGATA TAAATATCAA TATATTAAAT TAGATTTTGC
2641 ATAAAAAACA GACTACATAA TACTGTAAAA CACAACATAT CCAGTCACTA TGGCGGCCGC
2701 ATTAGGCACC CCAGGCTTTA CACTTTATGC TTCCGGCTCG TATAATGTGT GGATTTTGAG
2761 TTAGGATCCG TCGAGATTTT CAGGAGCTAA GGAAGCTAAA ATGGAGAAAA AAATCACTGG
2821 ATATACCACC GTTGATATAT CCCAATGGCA TCGTAAAGAA CATTTTGAGG CATTTCAGTC
2881 AGTTGCTCAA TGTACCTATA ACCAGACCGT TCAGCTGGAT ATTACGGCCT TTTTAAAGAC
2941 CGTAAAGAAA AATAAGCACA AGTTTTATCC GGCCTTTATT CACATTCTTG CCCGCCTGAT
3001 GAATGCTCAT CCGGAATTCC GTATGGCAAT GAAAGACGGT GAGCTGGTGA TATGGGATAG
3061 TGTTCACCCT TGTTACACCG TTTTCCATGA GCAAACTGAA ACGTTTTCAT CGCTCTGGAG
3121 TGAATACCAC GACGATTTCC GGCAGTTTCT ACACATATAT TCGCAAGATG TGGCGTGTTA
3181 CGGTGAAAAC CTGGCCTATT TCCCTAAAGG GTTTATTGAG AATATGTTTT TCGTCTCAGC
3241 CAATCCCTGG GTGAGTTTCA CCAGTTTTGA TTTAAACGTG GCCAATATGG ACAACTTCTT
3301 CGCCCCCGTT TTCACCATGG GCAAATATTA TACGCAAGGC GACAAGGTGC TGATGCCGCT
3361 GGCGATTCAG GTTCATCATG CCGTTTGTGA TGGCTTCCAT GTCGGCAGAA TGCTTAATGA
3421 ATTACAACAG TACTGCGATG AGTGGCAGGG CGGGGCGTAA AGATCTGGAT CCGGCTTACT
3481 AAAAGCCAGA TAACAGTATG CGTATTTGCG CGCTGATTTT TGCGGTATAA GAATATATAC
3541 TGATATGTAT ACCCGAAGTA TGTCAAAAAG AGGTGTGCTA TGAAGCAGCG TATTACAGTG
3601 ACAGTTGACA GCGACAGCTA TCAGTTGCTC AAGGCATATA TGATGTCAAT ATCTCCGGTC
3661 TGGTAAGCAC AACCATGCAG AATGAAGCCC GTCGTCTGCG TGCCGAACGC TGGAAAGCGG
3721 AAAATCAGGA AGGGATGGCT GAGGTCGCCC GGTTTATTGA AATGAACGGC TCTTTTGCTG
3781 ACGAGAACAG GGACTGGTGA AATGCAGTTT AAGGTTTACA CCTATAAAAG AGAGAGCCGT
3841 TATCGTCTGT TTGTGGATGT ACAGAGTGAT ATTATTGACA CGCCCGGGCG ACGGATGGTG
3901 ATCCCCCTGG CCAGTGCACG TCTGCTGTCA GATAAAGTCT CCCGTGAACT TTACCCGGTG
3961 GTGCATATCG GGGATGAAAG CTGGCGCATG ATGACCACCG ATATGGCCAG TGTGCCGGTC
4021 TCCGTTATCG GGGAAGAAGT GGCTGATCTC AGCCACCGCG AAAATGACAT CAAAAACGCC
4081 ATTAACCTGA TGTTCTGGGG AATATAAATG TCAGGCTCCC TTATACACAG CCAGTCTGCA
4141 GGTCGACCAT AGTGACTGGA TATGTTGTGT TTTACAGTAT TATGTAGTCT GTTTTTTATG
4201 CAAAATCTAA TTTAATATAT TGATATTTAT ATCATTTTAC GTTTCTCGTT CAGCTTTCTT
4261 GTACAAAGTG GTTGATATCC AGCACAGTGG CGGCCGCTCG AGTCTAGAGG GCCCGCGGTT
4321 CGAAGGTAAG CCTATCCCTA ACCCTCTCCT CGGTCTCGAT TCTACGCGTA CCGGTTAGTA
4381 ATGATCGACA ATCAACCTCT GGATTACAAA ATTTGTGAAA GATTGACTGG TATTCTTAAC
4441 TATGTTGCTC CTTTTACGCT ATGTGGATAC GCTGCTTTAA TGCCTTTGTA TCATGCTATT
4501 GCTTCCCGTA TGGCTTTCAT TTTCTCCTCC TTGTATAAAT CCTGGTTGCT GTCTCTTTAT
4561 GAGGAGTTGT GGCCCGTTGT CAGGCAACGT GGCGTGGTGT GCACTGTGTT TGCTGACGCA
4621 ACCCCCACTG GTTGGGGCAT TGCCACCACC TGTCAGCTCC TTTCCGGGAC TTTCGCTTTC
4681 CCCCTCCCTA TTGCCACGGC GGAACTCATC GCCGCCTGCC TTGCCCGCTG CTGGACAGGG
4741 GCTCGGCTGT TGGGCACTGA CAATTCCGTG GTGTTGTCGG GGAAGCTGAC GTCCTTTCCA
4801 TGGCTGCTCG CCTGTGTTGC CACCTGGATT CTGCGCGGGA CGTCCTTCTG CTACGTCCCT
4861 TCGGCCCTCA ATCCAGCGGA CCTTCCTTCC CGCGGCCTGC TGCCGGCTCT GCGGCCTCTT
4921 CCGCGTCTTC GCCTTCGCCC TCAGACGAGT CGGATCTCCC TTTGGGCCGC CTCCCCGCCT
4981 GGCGATGGTA CCGGTGTGGA AAGTCCCCAG GCTCCCCAGC AGGCAGAAGT ATGCAAAGCA
5041 TGCATCTCAA TTAGTCAGCA ACCAGGTGTG GAAAGTCCCC AGGCTCCCCA GCAGGCAGAA
5101 GTATGCAAAG CATGCATCTC AATTAGTCAG CAACCATAGT CCCGCCCCTA ACTCCGCCCA
5161 TCCCGCCCCT AACTCCGCCC AGTTCCGCCC ATTCTCCGCC CCATGGCTGA CTAATTTTTT
5221 TTATTTATGC AGAGGCCGAG GCCGCCTCTG CCTCTGAGCT ATTCCAGAAG TAGTGAGGAG
5281 GCTTTTTTGG AGGCCTAGGC TTTTGCAAAA AGCTCCCGGG AGCTTGTATA TCCATTTTCG
5341 GATCTGATCA GCACGTGTTG ACAATTAATC ATCGGCATAG TATATCGGCA TAGTATAATA
5401 CGACAAGGTG AGGAACTAAA CCATGGCCAA GCCTTTGTCT CAAGAAGAAT CCACCCTCAT
5461 TGAAAGAGCA ACGGCTACAA TCAACAGCAT CCCCATCTCT GAAGACTACA GCGTCGCCAG
5521 CGCAGCTCTC TCTAGCGACG GCCGCATCTT CACTGGTGTC AATGTATATC ATTTTACTGG
5581 GGGACCTTGT GCAGAACTCG TGGTGCTGGG CACTGCTGCT GCTGCGGCAG CTGGCAACCT
5641 GACTTGTATC GTCGCGATCG GAAATGAGAA CAGGGGCATC TTGAGCCCCT GCGGACGGTG
5701 CCGACAGGTG CTTCTCGATC TGCATCCTGG GATCAAAGCC ATAGTGAAGG ACAGTGATGG
5761 ACAGCCGACG GCAGTTGGGA TTCGTGAATT GCTGCCCTCT GGTTATGTGT GGGAGGGCTA
5821 AGCACAATTC GAGCTCGGTA CCTTTAAGAC CAATGACTTA CAAGGCAGCT GTAGATCTTA
5881 GCCACTTTTT AAAAGAAAAG GGGGGACTGG AAGGGCTAAT TCACTCCCAA CGAAGACAAG
5941 ATCTGCTTTT TGCTTGTACT GGGTCTCTCT GGTTAGACCA GATCTGAGCC TGGGAGCTCT
6001 CTGGCTAACT AGGGAACCCA CTGCTTAAGC CTCAATAAAG CTTGCCTTGA GTGCTTCAAG
6061 TAGTGTGTGC CCGTCTGTTG TGTGACTCTG GTAACTAGAG ATCCCTCAGA CCCTTTTAGT
6121 CAGTGTGGAA AATCTCTAGC AGTAGTAGTT CATGTCATCT TATTATTCAG TATTTATAAC
6181 TTGCAAAGAA ATGAATATCA GAGAGTGAGA GGAACTTGTT TATTGCAGCT TATAATGGTT
6241 ACAAATAAAG CAATAGCATC ACAAATTTCA CAAATAAAGC ATTTTTTTCA CTGCATTCTA
6301 GTTGTGGTTT GTCCAAACTC ATCAATGTAT CTTATCATGT CTGGCTCTAG CTATCCCGCC
6361 CCTAACTCCG CCCATCCCGC CCCTAACTCC GCCCAGTTCC GCCCATTCTC CGCCCCATGG
6421 CTGACTAATT TTTTTTATTT ATGCAGAGGC CGAGGCCGCC TCGGCCTCTG AGCTATTCCA
6481 GAAGTAGTGA GGAGGCTTTT TTGGAGGCCT AGGGACGTAC CCAATTCGCC CTATAGTGAG
6541 TCGTATTACG CGCGCTCACT GGCCGTCGTT TTACAACGTC GTGACTGGGA AAACCCTGGC
6601 GTTACCCAAC TTAATCGCCT TGCAGCACAT CCCCCTTTCG CCAGCTGGCG TAATAGCGAA
6661 GAGGCCCGCA CCGATCGCCC TTCCCAACAG TTGCGCAGCC TGAATGGCGA ATGGGACGCG
6721 CCCTGTAGCG GCGCATTAAG CGCGGCGGGT GTGGTGGTTA CGCGCAGCGT GACCGCTACA
6781 CTTGCCAGCG CCCTAGCGCC CGCTCCTTTC GCTTTCTTCC CTTCCTTTCT CGCCACGTTC
6841 GCCGGCTTTC CCCGTCAAGC TCTAAATCGG GGGCTCCCTT TAGGGTTCCG ATTTAGTGCT
6901 TTACGGCACC TCGACCCCAA AAAACTTGAT TAGGGTGATG GTTCACGTAG TGGGCCATCG
6961 CCCTGATAGA CGGTTTTTCG CCCTTTGACG TTGGAGTCCA CGTTCTTTAA TAGTGGACTC
7021 TTGTTCCAAA CTGGAACAAC ACTCAACCCT ATCTCGGTCT ATTCTTTTGA TTTATAAGGG
7081 ATTTTGCCGA TTTCGGCCTA TTGGTTAAAA AATGAGCTGA TTTAACAAAA ATTTAACGCG
7141 AATTTTAACA AAATATTAAC GCTTACAATT TAGGTGGCAC TTTTCGGGGA AATGTGCGCG
7201 GAACCCCTAT TTGTTTATTT TTCTAAATAC ATTCAAATAT GTATCCGCTC ATGAGACAAT
7261 AACCCTGATA AATGCTTCAA TAATATTGAA AAAGGAAGAG TATGAGTATT CAACATTTCC
7321 GTGTCGCCCT TATTCCCTTT TTTGCGGCAT TTTGCCTTCC TGTTTTTGCT CACCCAGAAA
7381 CGCTGGTGAA AGTAAAAGAT GCTGAAGATC AGTTGGGTGC ACGAGTGGGT TACATCGAAC
7441 TGGATCTCAA CAGCGGTAAG ATCCTTGAGA GTTTTCGCCC CGAAGAACGT TTTCCAATGA
7501 TGAGCACTTT TAAAGTTCTG CTATGTGGCG CGGTATTATC CCGTATTGAC GCCGGGCAAG
7561 AGCAACTCGG TCGCCGCATA CACTATTCTC AGAATGACTT GGTTGAGTAC TCACCAGTCA
7621 CAGAAAAGCA TCTTACGGAT GGCATGACAG TAAGAGAATT ATGCAGTGCT GCCATAACCA
7681 TGAGTGATAA CACTGCGGCC AACTTACTTC TGACAACGAT CGGAGGACCG AAGGAGCTAA
7741 CCGCTTTTTT GCACAACATG GGGGATCATG TAACTCGCCT TGATCGTTGG GAACCGGAGC
7801 TGAATGAAGC CATACCAAAC GACGAGCGTG ACACCACGAT GCCTGTAGCA ATGGCAACAA
7861 CGTTGCGCAA ACTATTAACT GGCGAACTAC TTACTCTAGC TTCCCGGCAA CAATTAATAG
7921 ACTGGATGGA GGCGGATAAA GTTGCAGGAC CACTTCTGCG CTCGGCCCTT CCGGCTGGCT
7981 GGTTTATTGC TGATAAATCT GGAGCCGGTG AGCGTGGGTC TCGCGGTATC ATTGCAGCAC
8041 TGGGGCCAGA TGGTAAGCCC TCCCGTATCG TAGTTATCTA CACGACGGGG AGTCAGGCAA
8101 CTATGGATGA ACGAAATAGA CAGATCGCTG AGATAGGTGC CTCACTGATT AAGCATTGGT
8161 AACTGTCAGA CCAAGTTTAC TCATATATAC TTTAGATTGA TTTAAAACTT CATTTTTAAT
8221 TTAAAAGGAT CTAGGTGAAG ATCCTTTTTG ATAATCTCAT GACCAAAATC CCTTAACGTG
8281 AGTTTTCGTT CCACTGAGCG TCAGACCCCG TAGAAAAGAT CAAAGGATCT TCTTGAGATC
8341 CTTTTTTTCT GCGCGTAATC TGCTGCTTGC AAACAAAAAA ACCACCGCTA CCAGCGGTGG
8401 TTTGTTTGCC GGATCAAGAG CTACCAACTC TTTTTCCGAA GGTAACTGGC TTCAGCAGAG
8461 CGCAGATACC AAATACTGTT CTTCTAGTGT AGCCGTAGTT AGGCCACCAC TTCAAGAACT
8521 CTGTAGCACC GCCTACATAC CTCGCTCTGC TAATCCTGTT ACCAGTGGCT GCTGCCAGTG
8581 GCGATAAGTC GTGTCTTACC GGGTTGGACT CAAGACGATA GTTACCGGAT AAGGCGCAGC
8641 GGTCGGGCTG AACGGGGGGT TCGTGCACAC AGCCCAGCTT GGAGCGAACG ACCTACACCG
8701 AACTGAGATA CCTACAGCGT GAGCTATGAG AAAGCGCCAC GCTTCCCGAA GGGAGAAAGG
8761 CGGACAGGTA TCCGGTAAGC GGCAGGGTCG GAACAGGAGA GCGCACGAGG GAGCTTCCAG
8821 GGGGAAACGC CTGGTATCTT TATAGTCCTG TCGGGTTTCG CCACCTCTGA CTTGAGCGTC
8881 GATTTTTGTG ATGCTCGTCA GGGGGGCGGA GCCTATGGAA AAACGCCAGC AACGCGGCCT
8941 TTTTACGGTT CCTGGCCTTT TGCTGGCCTT TTGCTCACAT GTTCTTTCCT GCGTTATCCC
9001 CTGATTCTGT GGATAACCGT ATTACCGCCT TTGAGTGAGC TGATACCGCT CGCCGCAGCC
9061 GAACGACCGA GCGCAGCGAG TCAGTGAGCG AGGAAGCGGA AGAGCGCCCA ATACGCAAAC
9121 CGCCTCTCCC CGCGCGTTGG CCGATTCATT AATGCAGCTG GCACGACAGG TTTCCCGACT
9181 GGAAAGCGGG CAGTGAGCGC AACGCAATTA ATGTGAGTTA GCTCACTCAT TAGGCACCCC
9241 AGGCTTTACA CTTTATGCTT CCGGCTCGTA TGTTGTGTGG AATTGTGAGC GGATAACAAT
9301 TTCACACAGG AAACAGCTAT GACCATGATT ACGCCAAGCG CGCAATTAAC CCTCACTAAA
9361 GGGAACAAAA GCTGGAGCTG CAAGCTT
//